Please read full Prescribing Information, including Boxed WARNING.
Please read full Prescribing Information, including Boxed WARNING.
Please read full Prescribing
Information, including Boxed WARNING.
The tools and documentation here can be important resources in securing treatment for your patients and getting them started on that treatment right away.
Check NUPLAZID approvals by your patient’s insurance provider to get a better sense of the cost and coverage options available to them.
Complete prior authorization that may be necessary to secure NUPLAZID coverage with your patient’s insurance provider.
LETTER OF APPEAL: Your patient may need a letter of appeal if a previous authorization request has been denied.
LETTER OF MEDICAL NECESSITY: You may need to document a patient’s medical necessity for treatment to get insurance coverage.
The following downloadable tools can help you more easily prescribe NUPLAZID and introduce patients and caregivers to the benefits of Acadia Connect.

Your patient’s NUPLAZID prescription may only
be filled at one of the following pharmacies*:

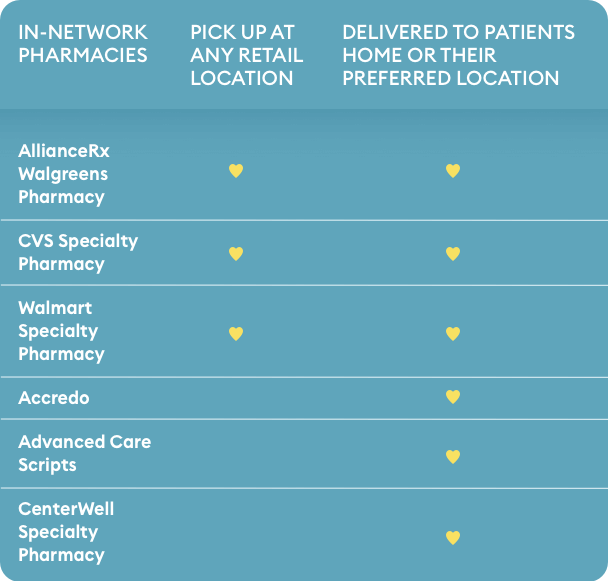
*If another pharmacy is selected, the prescription may not be filled.
Please read full Prescribing Information, including Boxed WARNING.
To start these services for you and your patients, complete our simple online prescription and enrollment form.
START ENROLLMENTIndication
NUPLAZID is indicated for the treatment of hallucinations and delusions associated with Parkinson’s disease psychosis.
Dosage and Administration
Recommended dose: 34 mg capsule taken orally once daily, without titration.
NUPLAZID is available as 34 mg capsules and 10 mg tablets.
Please read the full Prescribing Information, including Boxed WARNING.